Can you remember when the hottest trend in the healthiness of food was antioxidants, particularly antioxidant vitamins? It doesn’t seem that it was that long ago, but it is hard to find a recent article on the internet touting their benefits such as slowing down the aging process. In a quick search I only found one that has appeared in 2018. I have had an interesting interaction with oxidation in foods over the years. Before antioxidants became cool, I was working on lipid oxidation in certain fruits, like tomatoes and bell peppers, that lost quality when stored at normal refrigeration temperatures (1). The syndrome is known as chilling injury. When funding became available for oxidative damage to fruit and vegetable tissue, I had unfortunately moved on to other research topics and no longer had access to the scientific instruments I needed to continue that work.
The up-and-down interest in antioxidants illustrates the fickle nature of health trends in this country. Antioxidants are neither more nor less potent now than when they were popular a few years ago. Carefully controlled studies in model systems and animals showed their potential benefits. Large-scale studies with antioxidant supplementation did not live up to the promise shown in the earlier studies. The hype went away and so did sales of relevant pills and potions. Fruits high in antioxidants such as acai, blueberries and kiwifruit are still touted as critical parts of a healthy diet even though there is not much data available to make the case either way. Meanwhile, Americans continue to oxidize and continue to age.
Free radical theory of aging and other implications
Organisms build up free radicals as we age. A free radical contains an unpaired electron and thus becomes very reactive, particularly with oxygen. The resulting process is called peroxidation and the lipids (technical term for fats) are susceptible. Around every cell in the body is a membrane containing lipids and proteins. Membranes are also found within each cell. The lipids provide the membrane a barrier for cellular integrity—keeping in key components while preventing entry for those that do not belong. The proteins perform specific functions within a membrane such as enzymes to help in metabolism or as ports to selectively control passage of molecules from one side of the membrane to the other. Peroxidation of the lipids in the membranes can disable the structure to make it leaky or damage the proteins and reduce their ability to function properly.
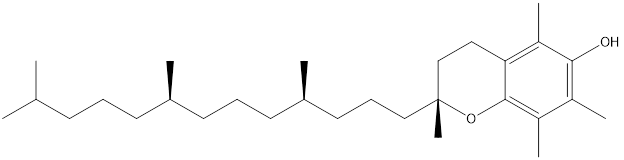 alpha-tocopherol (a form of Vitamin E)
alpha-tocopherol (a form of Vitamin E)
Fortunately, each cell in our bodies has defense and repair systems that can protect these delicate membranes from damage. The membrane is not a rigid structure but fluid. A cellular membrane has been likened to protein ships sailing in a sea of lipid. Fatty acids within the membrane lipids are not static as they move around from side to side and spin. An oxidized fatty acid can start a chain reaction that oxidizes its nearest neighbors. Antioxidants come to the defense of the membrane lipids before they can be broken down into toxic substances. The most effective antioxidant out there is alpha-tocopherol, a form of vitamin E that can be recycled in the liver and be reused. The tocopherol molecule becomes preferentially oxidized and stops the reaction. Then the repair mechanism can kick in as ascorbic acid (vitamin C) must be oxidized to regenerate the tocopherol. The ascorbic acid can be regenerated if there are enough reducing equivalents from flavonoid compounds found in green tea, blueberries and other fruits. The damaged fatty acids can be removed and replaced.
The above description is only one avenue of peroxidation that can occur in our bodies and other living organisms (2). It would appear that stopping the development of free radicals in the first place would be the best way to go. Many critical processes in nature, however, involve oxidation. In plants, photosynthesis requires chemical reactions that involve the formation of free radicals. In animals including ourselves free radicals are generated during metabolism and immune reactions. Thus, stopping all oxidative reactions in a cell could have serious consequences.
Maintaining health, then, is a balance between generating free radicals and protecting susceptible tissue from them. As we age, the damage due to free radicals begins to overtake our ability to defend and repair critical parts of our bodies. Peroxidized lipids can be rearranged, subdivided and modified by normal chemical processes in the body to form toxins. Many disorders in plants and animals involve oxidation. It is not always clear if these oxidative reactions are a cause or an effect of a specific disorder or merely part of its general progress.
Oxidation in foods
Lipids in foods are susceptible to oxidation. Generally speaking, the more fat and less water present in a product, the more likely that oxidation will occur. Polyunsaturated fats oxidize, but saturated and monounsaturated fats do not oxidize. Lipids in whole foods like nuts are just as likely to oxidize as those in formulated foods (those containing more than five ingredients). Foods containing polyunsaturated oils as ingredients or dried meat products are particularly susceptible to oxidation. To slow down oxidation in foods, antioxidants can be added. BHA (butylated hydroxyanisole) and BHT (butylated hydroxytoluene) are two artificial antioxidants that are particularly effective in preventing lipid oxidation in foods.

Molecular forms of BHA (butylated hydroxytoluene)
Tocopherols which are so effective in living tissues are not very useful in formulated foods. Oxidized lipids provide off-flavors sometimes producing odors such as grassy, paint-like and cardboard. Of more concern is that oxidized molecules formed during processing or storage of the finished product can be harmful to health. Oxidized fats in foods can be toxic (3). Food activists have called for the banning of artificial antioxidants in food products. Alternatively, I have heard members of Big Food describe BHA, BHT and similar molecules as cancer-preventing compounds. I like to think of these chemicals as an insurance policy that help prevent the accumulation of some serious toxins in our foods.
Precautions
We can consume antioxidants in the form of fruits and vegetables, fortified foods or supplements. If we are not able to protect our membranes with sufficient antioxidants we could become susceptible to natural pro-oxidants in our bodies, be less resistant to disease or even age prematurely. If we start to overdose on antioxidants, however, the antioxidant can turn into a pro-oxidant. At low levels, for example, alpha-tocopherol can have little or no effect. At higher levels, it can be a powerful protector of our membrane lipids. At even higher levels, however, it can accelerate peroxidation rather than preventing it. By trying to protect our bodies, we may actually be guilty of getting too much of a good thing!
Next week: Canned: The Rise and Fall of Consumer Confidence in the American Food Industry
(1) Shewfelt, R.L. and M.C. Erickson, 1991. Role of lipid peroxidation in the mechanism of membrane-associated disorders in edible plant tissue. Trends in Food Science & Technology, 2: 152-154.
(2) Niki, E., Y. Yoshida, Y. Saito and N. Noguchi. 2005. Lipid peroxidation: Mechanisms, inhibition and biological effects. Biochemical and Biophysical Research Communications 388:668-676.
(3) Viera, S.A., G. Zhang and E. Decker, 2017. Biological implications of lipid oxidation products. Journal of the American Oil Chemists’ Society 94: 339-351.
